International Journal of Scientific & Engineering Research, Volume 5, Issue 4, April-2014
ISSN 2229-5518
1592
University of Mosul
College of Science Dept. of Physics
Femtosecond laser plays an important role in cataract surgery, but retinal safety limits for near infrared laser employed in surgery are not well accuracy . This search determined retinal injury thresholds for protection from light scattering on bubbles and tissue fragments producing by laser cutting.
The aim of this research was to investigate possible retina damage under femtosecond (fs) - LASEK conditions and to optimize external parameters during surgery for reducing risk potential, such as pulse energy . Theoretically results indicated possible retinal damage, so this research tried to minimized these damages and get a good results theoretically.
![]()
An exceimer laser was used to ablate the
corneal surface tissue in the photorefractive keratomileusis (PRK) procedure (1). In 1990 the laser in situ keratomileusis (LASK) use a mechanical microtomecuts a thin flap in the corneal tissue (2) . Most of complications during LASK are caused by the fine cut of the microtome (3).Instead of mechanical devices, the cutting is done by focusing ultra- short laser pulses into the corneal tissue, these applications have been studied since its production (4). Studies about the risk potential of fs-LASK surgery were presented first in 2005 and 2006 (5-6) and have been continued and complemented within study. Over the last few years, international studies in femtosecond (fs) technology and its applications have been carried out concerning the side effects on the cornea and lens[7-10]..
In this research suppose that theoretically using laser of 10W, continuous wave of 1030 nm, with beam diameter of 2.6mm, so maximum that could be delivered to the eye was 3.6W.
The using of the common laser of control the power, a combination of half- wave by using a system of polarization. The retina damage thresholds under fs-LASIK conditions were determined dependent on varying laser parameters, such as irradiation time (tmin = 20 s, tmax = 150 s),numerical apertures (NA = 0.08, 0.23, 0.4 and 0.47) an dirradiation power (Pmin = 500 mW, Pmax =
1520mW).
IJSER © 2014 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 5, Issue 4, April-2014
ISSN 2229-5518
1593
The thermal properties of the tissue layer were derived based on the reported water content of each layer (11-13). Assuming that the heat capacity and thermal conductivity would be scattered average of the properties of water and hydrated proteins (14-15) which make up the reminder of material. The density of tissue was assumed to be that of
pure water fat(16) at 37C◦. The optical absorption coefficient for the retinal and choroid layers were taken from (17). Fig. (1-
2) show the relationship parameters of neural retina and retinal pigment.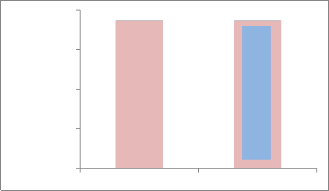
The Heat Capacity in (J/kg.K) is the same between Neutral retina and Retinal pigment epithelium, as shown in Fig. (3). As well as the Thermal Conductivity (Wm.K) is also the same as shown in Fig(4). That mean is the influence of temperature as the result of laser beam is the same.
4000
3000
2000
1000
0
1 2
Retinal pigment epithelium
100
80
60
40
20
0
1 2
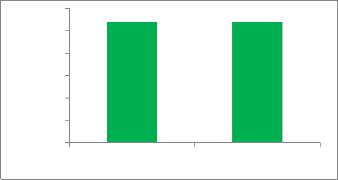
Neutral retina and Retinal pigment epithelium is the same.
0.6
0.5
0.4
0.3
0.2
0.1
0
1 2
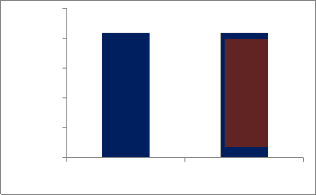
For the absorption of heat Fig.(5) shows that Retinal pigment will be absorbed more energy than Neutral retina ,it is very important for selected wavelength for minimize retinal damage .
IJSER © 2014 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 5, Issue 4, April-2014
100
80
1594
60 Neural
40
20
0
1 2
Temperature dynamics were calculated by numerically solving the bio-heat equation;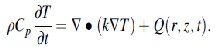 ------ (1)
------ (1)
In the equation, ρ is the density of the tissue, Cp is the heat capacity in the tissue layer, k is the thermal conductivity, and Q is the volumetric heat source term. The initial temperature was 37°C throughout the model and the boundaries of the computation domain had a fixed temperature condition at
37°C.Also we can say that laser heating is treated using Beer-Lambert law for absorption in non-scattering medium (17).
To study the damage of human eyes, should be describe the anatomy of the human eye by using the Gullstrand model(19)is used in determining the irradiance profile. During capsulotomy, the Gaussian laser beam is focused through water or tissue with similar refractive index with an NA of 0.1 (angle for 1∕e) on the anterior lens capsule, which is roughly 20.3 mm above the retina. For the 1030 nm wavelength, this
results in a beam radius of ∼1.5 mm on the
retina.
For calculating a conservative safety threshold power by assuming a stationary beam and applying the ANSI standard
following the retinal irradiance interpretation done by Delori et al.(20-21).
For the 1030 nm wavelength and retinal
permissible power P = 0.495t−0.25 W. From the typical pulse energy, pattern size, and spot spacing listed above, can calculate that the total energy E = 4.17 J is needed to form both capsulotomy and lens segmentation patterns. Assuming that the whole treatment is carried out with the same pulse energy and repetition rate, the fastest laser procedure that is within the ANSI safety limits can be
delivered in t = E∕P = (E/ 0.495(4∕3. For the total energy E listed above t = 17 s. The maximum average power P = E∕t is
then∼0.25 W and for the pulse energy of 6
μJ, the maximum repetition rate is
approximately 42 kHz. See Fig. (6).
18
16
14
12
10
8
6
4
2
0
The pulsed laser as a CW laser with the
same average power. The typical femtosecond laser in cataract surgery operates with repetition rates between 10 and
100 kHz, which translates to 10 to 100 μs between pulses. This speed makes the CW approximation valid for the crucial retinal and choroid layers because the beam radius
(∼1.3 mm) is large compared to the spot
IJSER © 2014 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 5, Issue 4, April-2014
ISSN 2229-5518
1595
spacing(5 to 10 μm) and the thermal
diffusion length for the time between pulses
(∼2–7 μm)(22).
There are two factors affect the optical
power reaching the retina a) plasma absorption and b) \\\bubble scattering. For attenuation plasma absorption and bubble scattering, can used a Ti: Sapphire(Tsunami, Spectra-Physics, Santa Clara, CA) femtosecond laser operating at 1 kHz with λ
= 800 nm and τ = 150 fs. Fig(7). A half-wave plate and polarizing beam splitter were used for attenuation. Responsible finally for scattering laser light from subsequent treatment scans can using gelatin or without using gelatin the residual bubbles trapped in the lens tissue, can scatter laser beam.
1200
1000
800
600
400
200
0
12
10
8
6
4
2
0
12
10
8
6
4
2
0
1 2
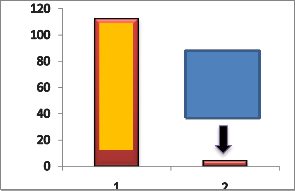
100s damage threshold is 11.7°C perfused and 12.6°C non perfused. Figures (10-11) .
30
Femtosecond laser.
25 23.8◦C
20
24.6◦ C
15 10 s
10
5
0
1 2 3
damage thresholds.
IJSER © 2014 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 5, Issue 4, April-2014
ISSN 2229-5518
1596
120
100
80
60
40
20
0
100
perfused
12.6 ◦ C
nonperfused
600
500
400
300
200
100
0
p min.= 152mW
100s damage thresholds.
a 1030-nm wavelength can be modeled safely
using Gaussian beam propagation although
shorter pulses may require. Otherwise less no
reaction will happen, more retina damage will be.
200
150
Tmin.= 20s

dependent on irradiation power maximum and power minimum.
Recently the study effects of femtosecond laser on retina are very important in the medical physics and ophthalmology. Thus in this research found that there are many parameters of femtosecond laser can be controlled to get a good results. First study the relationship between power total time and energy, Fig.(15)
20
15
10
100
50
0
1 2
0.5
5
0
more time it means that less power and energy.
0.4
0.3
0.2
0.1
0
NA=0.08
NA=0.23
frequency and wavelength depend upon minimum time in femtosecond as shown in Fig. (16).
1 2 3 4
IJSER © 2014 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 5, Issue 4, April-2014
ISSN 2229-5518
1200
1000
1597
800
600
400
200
0
1 2 3
10. Thomas RJ, Noojin GD, Stolarski DJ, Hall RT, Cain CP, Toth CA, et al. A comparative study of retinal effects from continuous wave and fem to second mode-lock lasers. Lasers SurgMed
2002; 31:1-17.
11. V. V. Tuchin et al., ―Eye tissues study,‖ Proc. SPIE 4427, 41–46 (2001).
12. R. S. Kadam and U. B. Kompella, ―Influence
frequency and wavelength depend upon minimum time in femtosecond.
1.Trokel SL, Srinivasan R, Braren B. Excimer
laser surgery of cornea. Am JOphtahlmol 1983;
2. Pallikaris IG, Papatzanaki ME, Stathi EZ, Frenschock O, Gerogiadis A. Laserin situ
keratomileusis. Lasers Surg Med 1990; 10(5):463-8.
3. Knorz MC, Jendritza B, Hugger P, Liermann A. Komplicationen der Laser-InSitu- Keratomileusis (LASIK). Der Ophtahlmologe
1999; 96:503-8.
4.König K, Riemann I, Stracke F, Le Harzic R. Nanoprocessing with nanojoulenear-infrared femtosecond laser pulses. Med Las Appl 2005;
5. Schumacher S, Sander M, Stolte A, Döpke c, Lubatschowski H. Investigationof possible fs- LASIK induced retinal damage surgery. Proc
SPIE 2006; 6318:344-52.
6. Schumacher S, Sander M, Stolte A, Döpke C, Gröne A, Ertmer W, et alInvestigation of retinal damage during refractive surgery. Proc SPIE
2005;5688:268-77.7
7. Rockwell BA, Cain CP, Roach WP, Thomas
RJ. Safe use of ultrashort lasers.Proc SPIE 1999;
8. Cain CP, Toth CA, Noojin GD, Carothers V, Stolarski DJ, Rockwell BA.Thresholds for visible lesiosn in the primate eye produced by ultrashortnearinfraredlaser pulses. Invest Ophthalmol 1999; 40(10):2343-9.
9. Cain CP, Toth CA, Noojin GD, Stolarski DJ, Thomas RJ, Cora S. et al. Visiblelesion threshold dependence on retinal spot size for femtosecond
laser pulses. JLaser Appl 2001; 13(3):125-31.
of lipophilicity on drug partitioning into sclera, choroid-retinal pigment epithelium, retina, trabecular meshwork, and optic nerve.,‖ J. Pharm. Exper. Ther. 332(3), 1107–1120 (2010).
13. H. Davson, ―The hydration of the cornea,‖
Biochem. J. 59(1), 24–28(1955).
14. P. H. Yang and J. A. Rupley, ―Protein-water interactions. Heat capacity of the lysozyme-water
system,‖ Biochem.18(12), 2654–2661 (1979).
15. A. Lervik et al., ―Heat transfer in protein-
water interfaces,‖ Phys. Chem. Chem. Phy.:
PCCP
16. V. Singh et al., ―On the thermal elevation of a
60-electrode epiretinal prosthesis for the blind,
‖ IEEE Trans. Biomed. Cir. Sys.2(4), 289–300 (2008).
17. M. Hammer et al., ―Optical properties of ocular fundus tissues—an in-integrating-sphere
technique and inverse Monte Carlo simulation,‖
Phys. Med. Biol. 40(6), 963–978 (1995).
18. J. Kandulla et al., ―Noninvasive optoacoustic
online retinal temperature determination during continuous-wave laser irradiation,‖ J. Biomed
Opt. 11(4), 041111 (2006).
19. H. Gross, F. Blechinger, and B. Achtner,
―Human eye,‖ in Handbook of Optical Systems:
Vol. 4, Survey of Optical Instruments 4 pp. 3–87, Wiley-VCH, Weinheim, Germany (2008).
20. ANSI, American National Standard for Safe
Use of Lasers, ANSI Z136.1-2007, Laser Institute
of America, Orlando, FL (2007).
21. F. C. Delori, R. H. Webb, and D. H. Sliney,
―Maximum permissible exposures for ocular safety (ANSI 2000), with emphasis on
ophthalmic devices,‖ J. Opt. Soc. Amer. A, Opt. Image Sci. Vis. 24(5), 1250–1265(2007).
22. M. Niemz, Laser-Tissue Interactions: Fundamentals and Applications, 2nd ed., Springer-Verlag, Berlin (2002).
IJSER © 2014 http://www.ijser.org