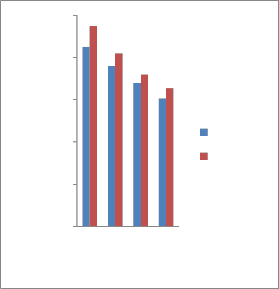International Journal of Scientific & Engineering Research, Volume 5, Issue 4, April-2014 1522
ISSN 2229-5518
THE INFLUANCE OF GARLIC (Allium sativum) OIL AS QUANCHING MEDIUM FOR HEAT TREATMENT OF PLAIN CARBON STEEL.
Sani. A. Salihu
Abstract
In this work, the garlic oil was evaluated as an alternative quench medium for hardening of plain carbon steel. The steel was heated to 860oC and socked for an hr, quenched and tempered separately in water and garlic oil. The mechanical properties (strengths, hardness, impact and ductility) of plain carbon steel (0.42%C) were investigated under two different quenching media (water and garlic oil). The investigations centered on water and garlic oil quenched and tempered conditions (3000C, 3500C, 4000C and 4500C). The results show that tensile strength of 2706.8
N/mm2 was obtained from sample quenched and tempered at 300°C in water, while garlic oil
gave lowest tensile strength value of 1906.9 N/mm2 when tempered at 450°C but the highest impact strength of 28 J. .The highest hardness value of 52.2 HRA was obtained from the sample quenched in water and tempered at 300°C, while the lowest hardness value of 41.6 HRA was also obtained from sample quenched in garlic oil and tempered at 450°C. There were variations in microstructure of tempered samples from the two quenchants used in this study. The tensile strength and hardness values were decreasing while impact strength and ductility appreciably increase with increasing of tempering temperature due to the decomposition of martensite. Result indicated that water quenched steel produced its best properties in strength and hardness, while garlic oil quenched steel has its best property in impact strength and ductility.
Keywords: Microstructure, Martensite, Tensile Strength, Hardness, Quenchants

Sani A. Salihu was a former Dean, School of Industrial Engineering, College of Engineering, and currently is the Coordinator, Renewable Energy Research Centre, Waziri Umaru Fed. Polytechnic, B/Kebbi, Kebbi State, Nigeria, Tel.+2347068433514. E-mail: sani.aliero@gmail.com
IJSER © 2014 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 5, Issue 4, April-2014 1523
ISSN 2229-5518
INTRODUCTION.
Steel is one of the most commonly used materials in industrial applications due to its low cost, high strength, and durability. Steel can be define as an alloy of iron and carbon usually containing between 0.015% to 2.14% carbon, often with other constituents such as manganese, silicon, sulphur, phosphorus (Gulyev, 1984, Higgins, 1995). Plain carbon steels are widely used for many industrial applications and manufacturing because of their low cost and easy fabrication (Smith and Hashemi
2006). They are classified on the basis of their carbon content as their major alloying element is carbon into low carbon steel, medium carbon steel and high carbon steel. According to Rajan et al. (1988), steels with carbon content varying from 0.25% to
0.65% are classified as medium carbon, while those with carbon content less than
0.25% are termed low carbon. The carbon content of high carbon steels usually ranges within 0.65-1.5%. Hardness and other mechanical properties of plain carbon steels increase with the rise in concentration of carbon dissolved in austenite prior to quenching during hardening heat treatment (Rajan et al. 1988; Thelning 1984), which may be due to transformation of austenite
into martensite (Feng and Tahir 2008).
The choice of correct quenching medium plays an important role in determining the desire properties of engineering materials. The basic function of a quench medium is to control the rate of heat transfer from the surface of the part being quenched (Kulikov, 1997, Higgins,
2001). Accordingly, many components use oil quenching to achieve consistent and repeatable mechanical and metallurgical properties and predictable distortion patterns. The reason oil quenching is so popular is due to its excellent performance results and stability over a broad range of operating conditions. Oil quenching facilitates hardening of steel by controlling heat transfer during quenching, and it enhances wetting of steel during quenching to minimize the formation of undesirable thermal and transformational gradients which may lead to increased distortion and cracking. Therefore, the mechanical strength of medium carbon steels can be improved by quenching in appropriate medium. However, the major influencing factors in the choice of the quenching medium are the kind of heat treatment, composition of the steel, the sizes and shapes of the parts (Rassizadehghani et al. 2006). As explained by Grishin and Churyukin (1986) , mineral
oils have been found to exhibit best cooling
IJSER © 2014 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 5, Issue 4, April-2014 1524
ISSN 2229-5518
capacity for the majority of alloy steels but they are relatively expensive, toxic and non- biodegradable.
Currently, vegetable oils are one of the most commonly identified renewable, biodegradable and non-toxic quenchants. In addition to their relatively narrow viscosity range, vegetable oils exhibit considerably lower thermal-oxidative stability relative to petroleum oil, which has been a significant factor inhibiting its use in the industry. As an alternative to mineral oil, vegetable oils are used as quench media. They are cheap, abundantly available, biodegradable, renewable and environment friendly (Tagaya and Tamura, 1954). There is an increasing interest in the use of vegetable oils as renewable, biodegradable and non- toxic alternatives to petroleum oil-based quenchants for the heat treating industry. One of the first commercial references to the use of vegetable oil as a metal quenchant was a patent issued in 1932 (Tagaya and Tamura, 1954). More recently, the use of locally available vegetable oils, which are relatively cheap, non-toxic and environmental friendly, as quenching media, has begun to generate attention (Ndaliman
2006).
In the present work, garlic oil is used as quenchant to study its effectiveness. Its widespread availability and low cost make garlic oil an ideal component for developing renewable, bio-based products.. After heating the steel it is quenched and tempered in tap water and one biodegradable quenchant i.e. garlic oil and tested for various mechanical properties.
2.0 RESEARCH METHODOLOGY Sample of plain carbon steel bar with 16 mm diameter and 2 m long was purchased from a local
market located at Jega, 50 kilometers away from Birnin Kebbi, Northern, Nigeria. The chemical composition of material under investigation is given in Table 1 while Table
2 is the physicochemical properties of garlic oil. Standard tensile and impact specimens were made from steel sample using lathe machine according to AISI standard. Samples were subjected to hardening, and tempering at four different tempering temperatures and were tested for tensile, yield, percentage elongation, hardness and impact toughness. Each test was conducted three times and the average taken represents the results recorded in Table 3.
IJSER © 2014 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 5, Issue 4, April-2014 1525
ISSN 2229-5518
Table 1. Chemical composition of steel used
Element | C | Mn | Si | S | P | Fe |
Composition (wt, %) | 0.42 | 0.63 | 0.37 | 0.001 | 0.002 | Bal |
Table 2. Some physicochemical properties of oils used
Type of oil | Colour | Specific gravity, (g) | Viscosity at 400C, (poise) | Flash point, (0C) | Iodine value | Free fatty acid, (% Oleic acid) | Boiling point, (0C) | Saponificati on value (MgKOH/g) |
Garlic | Light yellow | 0,95 | 0.098 | 145 | 12.73 | 2.10 | 140 | 193 |
2.1 Heat Treatment
Generally heat treatment is accomplished in three stages: Stage l—Heating the samples slowly to ensure a uniform temperature. Stage 2—Soaking (holding) the samples at a given temperature for a given time. Stage
3—Cooling the samples to room temperature (Rajan et al 1988). Specimen for heat treatment and subsequent hardness, impact and tensile testing were obtained from 20 mm diameter bar of a 0.42%C medium carbon steel. A muffle furnace was used for austenitizing the specimens at
8600C for 1 hr and then quenched separately
in tap water and garlic oil. Then the specimens were subjected to tempering heat treatments, within the temperature range of
300-4500C. After which the hardness, impact and tensile properties were determined in as tempered state using Rockwell hardness tester, Charpy impact tester and universal tensile testing machine respectively.
2.2 Mechanical Test
2.2.1 Tensile Testing
Tensile test were carried out on the quenched specimens using Instron Universal Tester. Each
of the specimens was loaded till fractured, and the fracture load for each sample was recorded as well as the diameter at the point of fracture and the final gauge length. The initial diameter and initial gauge length for
each sample was noted before the
IJSER © 2014 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 5, Issue 4, April-2014 1526
ISSN 2229-5518
application of the uniaxial load. The percentage elongation of each test sample was determined, as well as the tensile strength.
2.2.2 Hardness Test
For hardness testing, oxide layers formed during heat treatment were cleaned and polished. Rockwell hardness method was used for the determination of the hardness of the quenched and tempered samples. The specimens were brought in contact with the pyramid indenter and allowed to rest for a dwell time. The hardness of the specimens were indicated by the penetration of the indenter on the test specimens, and displayed by the machine. Average values were recorded after repeating the test for each of the test specimens.
2.2.3 Impact Test
Heat-treated samples were tested for various mechanical properties. Impact energy was
recorded using the Charpy impact tester. Charpy V-notch specimens were machined from tempered rod, in the ASTM standard form of 10mm×10mm×55mm. Both tensile, hardness and impact data reported in this work are the average values of the data obtained from steel samples. All testing was done in accordance with ASTM standard test procedures (ASM, 2008).
3.0 Results and discussion
The tensile, hardness and impact tests were performed at room temperature and the results of tensile, hardness, impact and percentage elongation tests are presented in Table 3. As observed, the both the ultimate tensile strength and hardness decrease with increase of tempering temperature for both quenchants. However, the impact strength and percentage elongation increase with increase of tempering temperature.
Table 3. Mechanical Properties of various samples used
Quenchant | Heat treatment | Tensile strength, N/mm2 | Yield strength, N/mm2 | Hardness, HRA | Impact strength, J | Percentage Elongation, % |
Garlic oil | Quenched at 8600C and tempered at 3000C Quenched at 8600C and tempered at 3500C Quenched at 8600C and tempered at 4000C Quenched at 8600C and | 2457 2173 1906 | 1942 1894 1696 | 51.3 45.2 41.6 | 18 19 23 | 9 12 15 |
IJSER © 2014 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 5, Issue 4, April-2014 1527
ISSN 2229-5518
| tempered at 4500C | 1732 | 1512 | 36.3 | 28 | 19 |
Water | Quenched at 8600C and tempered at 3000C Quenched at 8600C and tempered at 3500C Quenched at 8600C and tempered at 4000C Quenched at 8600C and tempered at 4500C | 2706.8 2385.3 2178.9 1954.2 | 2369.4 2046.6 1798.4 1635.2 | 53.2 51.3 48.5 45.9 | 11 13 17 19 | 1 3 6 8 |
3.1 Microstructure.
Metallographic examination was carried out on the medium carbon steel specimens. Standard techniques for specimens preparation for the microstructure observation were adopted. The samples were mounted on bakelite using silicon carbide abrasive papers (240 to 600 grits sizes). Following the polishing of specimens on a rotating disc and subsequent surface
cleaning, etching of the polished specimen was done using 2% Nital (2% Nitric acid, HN03 , in ethyl alcohol) for 15-30 seconds to develop the structure. The structures
obtained were photographed, and are shown in Figure 1.
3.1.1 Effect of quenching media on microstructure and mechanical properties
The quenching media under investigation have shown an effect on the microstructural
formation and mechanical properties of the
steel because of their different
physicochemical properties. Figure 1 (a). shows the microstructure of the as-received medium carbon steel which reveals the presence of ferrite, pearlite and inclusions. As explained by Novikov, (1980), different quenching media have significant role in determining the final structure of an alloy because of different cooling rates. The quenching media under investigation have different cooling rate . Of the quenching media under investigation, water is the most severe and garlic oil the least. Figure 1
shows microstructures of two specimens that underwent the same heat treatment conditions, but with different quenching media. The results of structural formations show different proportions of martensites, retained austenite and carbides. Figure 1
also show that with increase of tempering temperature, structural transformations were
observed revealing more bainitic structure,
IJSER © 2014 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 5, Issue 4, April-2014 1528
ISSN 2229-5518
less retained austenite for samples tempered in garlic oil and martensite more especially with samples that were tempered at 300 to
3500C in water.
When the tempering temperature is higher than 3500C the cementite in the tempered martensite coarse as explained by Lakhtin, (1977). With an increase in the tempering temperature the carbon concentration of the matrix in the tempered martensite still
decreases due to the diffusion of carbon atoms to the cementite. The strength of the tempered martensite decreases and its ductility increases (Senthilkumar and Ajiboye, 2012). Therefore, the ultimate tensile strength of the tempered ferrite– martensite decreases and its elongation increases as observed by Senthilkumar and Ajiboye, (2012), and Ashish Vermaa, Pravin Kumar Singh, (2013). When the specimens (garlic oil) are tempered at temperature
450℃, phase transformation takes place
from retained austenite to bainite. Bainitic
structure consist of ferrite and epsilon carbide which are formed at temperature
ranging between 400℃−450℃.


C) d)


e) f)


g) h)

i)


a) b)
IJSER © 2014 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 5, Issue 4, April-2014 1529
ISSN 2229-5518
Figure 1. Microstructures of quenched and tempered showing the distribution of tempered carbides: a) as-quench; shows pearlite in matrix ferrite, b) QWT; 3000C, c)
QOT; 3000C, d) QWT; 3500C, e) QOT;
3500C, f) QWT; 4000C, g) QOT; 4000C, QWT; 4500C, QOT; 4500C
(QWT means quenched in water and tempered, while QOT is quenched in oil and
tempered).
TABLE 4: Microstructure of Quenched and Tempered Steel in Two Quenchants
QUENCHING MEDIA AND TEMPERING TEMPERATURE | MICROSTRUCTURE | |
Untreated sample | Ferrite (white) and pearlite (dark) | |
Water - 3000C (QWT) | Ferrite (white) and coarse carbide (dark) | |
Water – 3500C (QWT) | ferrite (white) and coarse carbide (dark) | |
Water – 4000C (QWT) | ferrite and coarse tempered martensite | |
Water – 4500C (QWT) | Consists of an equilibrium coarse mixture of ferrite (white) and carbide (dark) | |
Garlic oil – 3000C (QOT) | Fine tempered martensite plus ferrite | |
Garlic oil – 3500C (QOT) | Consists of coarse tempered martensite and bainite | |
Garlic oil – 4000C (QOT) | Coarse tempered martensite and bainite | |
Garlic oil - 4500C (QOT) | Tempered martensite and bainite | |
3.2 Mechanical properties
3.2.1 Ultimate tensile strength.
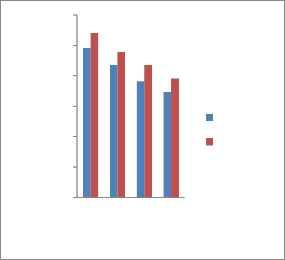
Fig. 2 shows the variation of ultimate tensile strength (UTS) of the garlic oil and tap water as a function of tempering temperature. At 300°C, sample quenched in
garlic oil displayed tensile strength of
2457/mm2 , while tap water exhibited highest tensile strength value of 2706.8
N/mm2. At 350°C, all the quenched and
tempered samples gave lower tensile strength value, with sample quenched in garlic oil showing lowest tensile strength
value because of the formation of tempered
IJSER © 2014 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 5, Issue 4, April-2014 1530
ISSN 2229-5518
martensite. However, at 400°C and 450°C, heat treated samples quenched in garlic oil displayed slight decrease of tensile strength because of decomposition of martensite.
The effect of tempering temperature on yield strength is illustrated in Fig. 3. Yield
3000
2500
2000
1500
Strength responded to the tempering temperature in same manner with Tensile Strength. Yield Strength continuously decreased with increasing tempering temperature for both samples quenched and tempered in garlic oil and water.
3.2.2 Variation of the hardness of the tempered samples
The hardness property of tempered
specimens is measured as function of
1000
500
0
300 350 400 450
Garlic oil
Tap water
tempering temperature using Rockwell
hardness tester. The results obtained from
testing of specimens quenched at 860℃ and
tempered at temperature 300, 350, 400 and
Tempering Temperature, 0C
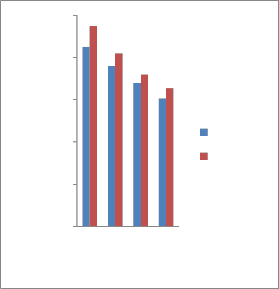
Fig. 2. Effect of tempering temperature on ultimate tensile strength for different quenchants
2500
2000
1500
Garlic oil
450℃ was shown in Fig. 4. There are two
different significant trends in this graph
because of changing of structural effect. The highest Rockwell hardness value of the samples quenched in tap water and garlic oil were obtained at 300°C, with peak value from the sample quenched in tap water, as shown in Figure 4. However, at 350°C, the hardness values of water the quenched
sample decreased slightly, with those
1000
500
0
300 350 400 450
Tap water
quenched in garlic oil became lower than that of the as-received sample, which is 42.3
HRA. The hardness values of all the samples
increased at 300°C above that of the as-
Tempering temperature, 0C
Fig.3 Effect of tempering temperature on yield strength for different quenchants
received, but later decreased when quenched after heating up to 450°C. Moreover, the resulting tempered structures are expressed
IJSER © 2014 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 5, Issue 4, April-2014 1531
ISSN 2229-5518
in Figure 1b to 1i. According to these microstructures, bainitic structure (ferrite and epsilon carbide) are formed at 400°C to
450°C. This indicates that the best heating temperature to obtain highest hardness value will be 300°C with garlic oil as quenching medium. There was decrease in hardness
60
50
40
30
Garlic oil
because of the formation of tempered martensite and bainite and structure when tempered in garlic oil, while the tempered samples quenched in tap water experienced slight decrease.
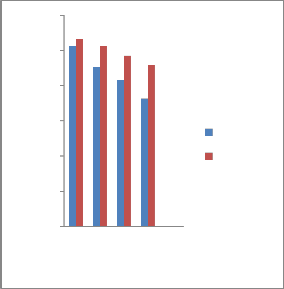
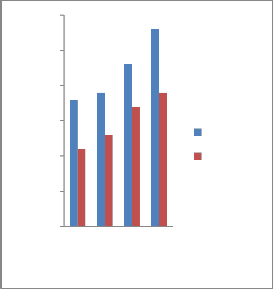
3.2.3 Impact properties
The mechanical properties of tempering samples (tempered at 3000C, 3500C, 4000C and 4500C) showed that the Toughness in J increases with increase of tempering
temperature. The results of the specimens which were hardened with tap water and
20
10
0
300 350 400 450
Tap water
garlic oil quenched and then tempered at
3000C, 3500C, 4000C and 4500C are expressed graphically in Figure.5. Impact toughness responds to tempering
Tempering temperature, 0C
Fig. 4. Variation of hardness against tempering temperature for different heat treatments
temperature in the similar manner to
percentage elongation.
IJSER © 2014 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 5, Issue 4, April-2014 1532
ISSN 2229-5518
30
25
20
15
10
5
0
300 350 400 450
Garlic oil
Tap water
3. The heat treatment of steel using garlic quenchant gives the better mechanical properties and microstructure by forming ferrite and epsilon carbide at 350°C to
400°C, hence garlic oil stands as a potential quenching medium in heat treatment of steel.
References:
Ashish Vermaa, Pravin Kumar Singh
Tempering temperature, 0 C
Fig. 5 Effect of tempering temperature on impact strength of specimens tempered in different quenchants.
4.0 Conclusions
The following conclusion has been drawn from the experimental result and discussion already made.
1. The results show that the ultimate tensile strength and the yield strength decrease, whereas the elongation and impact strength increase with increase in tempering temperature.
2. Ultimate tensile strength decreases continuously by increasing tempering temperature. The ductility of the specimen is measured by the tensile test. The elongation increases with the increase in tempering temperature.
(2013): Influence of Heat Treatment on
Mechanical Properties of Aisi1040 Steel, IOSR Journal of Mechanical and Civil Engineering (IOSR-JMCE), pp. 32-38
ASTM E18, (2008): Standard Test Method for Rockwell Hardness of Metallic Materials, American Society of Testing and Materials,
ASTM E8, (2008) Standard Test Method for Tension Testing of Metallic Materials, American Society of Testing and Materials, Feng, C.; and Tahir, I.K. 2008. The effect of quenching medium on the wear behaviour of a Ti-6Al-4V alloy. J. Mater. Sci. 43(2): 788-
92.
Grishin, S.A.; and Churyukin, Y.N. 1986. Evaluation of the cooling capacity of quenching media based on water. Metal Sci. Heat Treat. 28(10): 744-5.
Kulikov, A.I. 1997. A new quenching medium for metals and alloys. Metal Sci.
Heat Treat. 39(11-12): 528-30.
IJSER © 2014 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 5, Issue 4, April-2014 1533
ISSN 2229-5518
Lakhtin, Y.M. (1977). Engineering Physical Metallurgy and Heat Treatment. Mir Publishers, Moscow, USSR
Ndaliman, M.B. 2006. An assessment of mechanical properties of medium carbon steel under different quenching media. AU J.T. 10(2): 100-4.
Rajan, T.V.; Sharma, C.P.; and Sharma, A.
1988. Heat Treatment - Principles and Techniques. Prentice-Hall of India Private Limited, New Delhi, India
Senthilkumar, T.; and Ajiboye, T.K. 2012. Effect of heat treatment processes on the
mechanical properties of medium carbon
steel. Journal of Minerals and Materials
Characterization and Engineering: pp143-
152.
Smith, W.F.; and Hashemi, J. 2006. Foundations of Materials Science and Engineering. 4th ed. McGraw-Hill, Ney York, NY, USA.
Thelning, K.-E. 1984. Steel and Its Heat Treatment. 2nd ed. Butterworth, London, England, UK.
Tagaya, M., Tamura, I.,(1954) “Studies on the quenching media 3rd report, “The cooling ability of oils”. Technology Report,
Osaka University, Vol. 4, p. 305-319.
IJSER © 2014 http://www.ijser.org