International Journal of Scientific & Engineering Research, Volume 4, Issue 6, June-2013 3009
ISSN 2229-5518
Emission Control of DI Diesel Engine by Using Aque- ous Salt Solution
Md. Amzad Hossain, Md. Rasedul Islam, Md. Hasan Ali, Mohammad Mashud
Abstract— Vehicle emission control is the study & practice of reducing the motor vehicle emissions especially internal combustion engine. It includes a variety of engine management and emission control devices and system. They all work to reduce the air pollutants in the exhaust gas. Air pollution can be defined as addition to our atmosphere of any material which will have a deleterious effect on life upon our planet. The main pollutants contributes by automobiles are carbon monoxide (CO), unburned hydrocarbons (UBHC), oxides of nitrogen (NOx), smoke, odor, particulate matter(PM). Water oil stable emulsion and obtained a significant reduction of NOX and improved break spe- cific fuel consumption (BSFC). Noboru Miyamoto et al. examined the effect of several aqueous metal salt solutions on NOX reduction in a DI diesel engine. It was found that with sodium salt solution, NOX and smoke emission decreased by more than 60% and 50% respectively with respect to conventional operation. The effect of diesel/water emulsion and associated gaseous emission were Investigated by G.E Andrews et al. It was noticed that both NOX and particulate emission decreased with increased water content. The source of high ambient air ozone and particular levels include emissions from essentially all combustion devices, i.e., truck and bus diesel engines have been identified as a significant mobile source of both oxides of Nitrogen (NOX) and particulate matter (PM). It produces 50% of the total amount of NOx and HC emissions and as much as 80% to 90% of the CO an HC concentrations in urban air tend to rise. NOx and a precursor to ozone formation in the lower atmosphere and diesel particulate matters. So, it is the very urgent problem to reduce the exhaust emissions from diesel engine. Many development programs carried out such as engine design modification, treatment of exhaust gas, fuel modifica- tion. This experiment is used mainly for reducing NOX by using fuel modification system with aqueous salt solution. Here study also per- forms about other particulate emissions with various engine efficiency parameters.
Index Terms Vehicle emission control, aqueous salt solution, engine efficiency, formation of NOX.
—————————— ——————————
1 INTRODUCTION
IRECT injection (DI) diesel being to the group of internal combustion engine which has a long history since Lenoir first developed the spark ignition engine in 1860 and Ru-
dolph Diesel invented the compression ignition engine in
1892.Now a days the IC engine play a dominant role in the
fields of power, propulsion and energy. The last 30 to 40 years
have been an explosive growth in engine research and devel- opment based on the factors. Firstly, the need to control the automotive contribution to urban air pollution. Secondly, the need to achieve significant improvement in automotive fuel consumption. Thirdly, for increasing power.
————————————————
• Md. Amzad Hossain is Lecturer and currently pursuing masters degree in
Mechanical Engineering Departmentof Khulna University of Engineering
& Technology, Bangladesh. E-mail: amzad59@mail.com
• Md. Rasedul Islam is Lecturer in Mechanical Engineering Department of
Khulna University of Engineering & Technology, Bangladesh E-mail:
shihab_me.kuet@yahoo.com
• Md. Hasan Ali is Assistant Professor in Energy Technology Department
and currently pursuing masters degree in Mechanical Engineering De- partmentof Khulna University of Engineering & Technology, Bangladesh.
E-mail: mhakuet@yahoo.com
• Mohammad Mashud is Professor in Mechanical Engineering Department
of Khulna University of Engineering & Technology, Bangladesh. E-mail:
mdmashud@yahoo.com
Recent researchers have focused their attention to carbon dioxide (CO2) emissions, which are carcinogenic. The emission of CO2 is directly proportional to the consumption and is said to be responsible for future changes in world’s climate. The diesel engines, especially DI diesel engines are the most effi- cient use of fossil fuels for vehicle propulsion.
Fuel consumption and CO2 emissions from DI diesel en- gines are 20-30 percent less than gasoline engines. The merit implies less impact on climate change as well as saving of en- ergy resources. This is advantageous side of DI diesel engine and a reason of increasing in numbers worldwide. The un- burnt hydrocarbon (UHC) and carbon monoxide emissions are also significantly lower in diesel engines than that of gasoline engines without a catalyst. Moreover, diesel engines have the best reliability and durability over the other power plants. Perhaps more impressive is the durability of the diesel engine. It is not uncommon for diesel engine to have a life of 1,000,000 miles in heavy duty trucks, to power city buses for up to 15-20 years, and to power non road equipment for several thousand hours before requiring rebuild or replacement. However, higher particulate matters (PM) and nitrogen oxides (NOX) are the disadvantages.
Further, in some engine running conditions such as at idling or at low temperature operations, the exhaust odor from DI diesel engines is considerably higher than that from gasoline engines. Most of the diesel engine researchers have focused their attention to reduce PM and NOX, because these are the
IJSER © 2013 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 4, Issue 6, June-2013 3010
ISSN 2229-5518
regulated emissions. And for better marketability of diesel engine this emission must be reduced below to the regulated limit. After the introduction of the emissions standards in the
early 1960 in California, USA, there occurred a many-fold ef- fort in engine research. The odor reduction research was also started almost at the time and most of the significant diesel exhaust odor research has been conducted from the middle of
1960.
2 METHODOLOGY
Diesel engine belongs to the group of CI engine of the IC en- gine. Now a day the IC engine plays a dominant role in the fields of power, propulsion and energy. The links between public objection ability and the level of diesel exhaust emis- sion have been more or less established [1]. According to the M.L.Mathur and R.P.sharma it is found that especially NOx has adverse effect on human health. When the vehicles with the DI diesel were increased this effect becomes an emergence to all [3]. In the exhaust of internal combustion engines, NOx refers to a class of compounds called nitrogen oxides. In DI diesel engine exhaust, nitric oxide (NO) is usually the most abundant nitrogen oxide and constitutes more than 70-90% of total NOx. Nitrogen dioxide (NO2) is also present at signifi- cant levels.
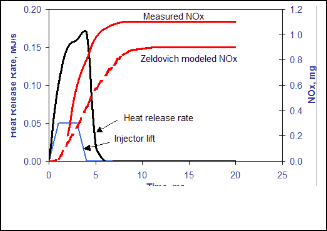
Figure 1 shows a measured time series profile of NOx for- mation that is typical of many diesel engine operating condi- tions. Rapid NOx formation begins well after the start of heat release. Shortly after the end of heat release, the period of rap- id NOx formation ends because temperatures of the burned gas decrease due to mixing with cool bulk gas and expansion of the charge.
Fig. 1. Profiles of Heat Release Rate from Combustion Bomb
Studies
Recently the emission researchers of diesel engines are going on mostly for reduction of regulated emissions such as NOx, PM, and other due to severe regulation standards of those components. Table 2 shows the quantity of emission per Kg.
The most effective odor reduction method is the fitment of minimum sac volume injectors in that large reduction of ex- haust odorant concentration are attainable with little or no
deterioration in thermal efficiency or smoke [10]. Further re- duction may be obtained by minimizing fuel dispersion dur- ing the ignition delay period.
TABLE 1
JAPANESE EMISSION STANDARDS FOR LIGHT DUTY DIESEL EN-
GINES
Year | NOx(g/km) | PM(g/km) | HC(g/km) | CO(g/km) |
1994 | 0.6 | 0.2 | 0.4 | 2.1 |
1998 | 0.4 | 0.08 | 0.4 | 2.1 |
2002 | 0.3 | 0.05 | 0.12 | 0.63 |
PM = Particulate matters; NOX = Nitrogen oxides: CO = Carbon Monooxide; HC =
But recent researches have shown that the above mentioned system are not so accurate and cannot able to reduce NOx to a satisfactory level. Main reduction techniques are as follows.
i. By optimizing the engine parameters.
ii. By using alternative fuels. iii. Exhaust gas after treatment
a. catalytic system
b. absorption system
Catalytic method is only applicable when the vehicle is in
running condition where the exhaust gas temperature is above
2500c or so. From the above discussion of odor reduction
techniques IT is found that, there are no appropriate methods
to reduce odor at low temperature condition. Hence this study focuses on odor reduction at low exhaust temperature condi- tion at idling by water washing an absorption type system.
2.1 Diesel Emission
To make any proper exhaust emission strategy, it is important first to know the combustion phenomena that influence the exhaust emission. The following is the generalized diesel combustion reaction,
CxHy(Sz)+(O2+N2)= CO2+H2O+(O2+N2)+(NOx+HC+OOC+C+CO+SOx)
The diesel combustion reaction consist of hydrocarbon chains (CxHy) being oxidized in an explosive reaction to form carbon dioxide (CO2) and water however, the reaction is not 100% efficient and the constituent are not pure. The air is used to supply the oxygen contains about 80% of nitrogen and the diesel fuel contains a small percentage of sulpher. The result is that trace of amount of either chemical are found in the reac- tion. All of the trace constituents are of concern to the envi- ronment or can pose a health risk in higher constituents. The main harmful pollutants that emit in diesel exhaust are:
IJSER © 2013 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 4, Issue 6, June-2013 3011
ISSN 2229-5518
a) Oxide of nitrogen (NOx) b) Particulate matter (PM) c) Carbon monoxide (CO)
d) Unburnt hydrocarbon (UBHC)
e) Odor
a) Oxide of Nitrogen (NOx):
Oxides of nitrogen are a combination of nitric oxide (NO) and nitrogen di oxide (NO2). The air supplied for combustion con- tains about 77% of nitrogen. At lower temperature the nitro- gen is inert but at temperature higher than 1100OC nitrogen reacts with oxygen. Therefore high temperature and availabil- ity of oxygen is available with highest local peak combustion temperature highest amount of NOx is formed in diesel ex- haust. The quantity of NOx varies from a few hundred to well over 1000 PPm in the diesel exhaust.
b) Particulate Matter (PM):
Organic and inorganic compound of higher molecular weight are exhausted in the form of small size particles of the older of
0.02 to 0.06 bul. Hundreds of separate organic compounds can be formed when the combustion reaction is not complete. These organic fractions (VOF). When the concentration of the organic compound in the exhaust rises, they can pose severe health risks.
c) Carbon Monoxide (CO):
It is product of incomplete combustion due to insufficient amount of air in the air fuel mixture. It is not formed in large quantities due to the excess amount of oxygen available dur- ing combustion and generally not the concern.
d) Hydrocarbons:
Unburned hydrocarbon emissions are direct result of incom- plete combustion.
e) Odor:
The odorous emissions are believed in the group of other or- ganic compound (OOC) and highly dependent on the combus- tion process which influence by the engine parameters and fuel consumptions. The products of partial oxidization are the main cause of odor in diesel exhaust.
Other harmful pollutant of sulpher di oxides is directly related
to the amount of sulphur in the diesel fuel. Higher Sox emis- sions can contribute to environment concern elated to the for- mation of acidic compounds in the atmosphere.
2.2 Cause of Pollutants Formation:
The various causes of these harmful pollutants are as follows:
a) Causes of NOx:
The two main reasons for the formation of NOx are high tem- perature and availability of oxygen. Engine design and the model of vehicle operation affect the NOx concentration in exhaust. A pre combustion chamber engine produces less NOx than the direct injection engine due to lower peak tem- perature. the maximum NOx is formed at ratios between
14:1and 16:1. At lean and rich air fuel mixtures the NOx con-
centration is comparatively low. At high fuel air ratio the addi- tional fuel tends to cool the change, so the localized peak tem- perature and lowered resulting in drop in NOx concentration. The NOx formation is also significantly affected by injected system and time. Also the variation in fuel characteristics such as cetane number viscosity modulus of elasticity and rate of burning, etc., all contribute to differences in NOx level ob- tained from different levels.
b) Causes of Smoke:
The cause of smoke is incomplete burning of fuel inside the combustion chamber. Two main reasons for incomplete com- bustion are incorrect air fuel ratio and improper mixing. These might result due to engine design factors, such as injection system characteristics, the induction system, governor control the fuel used and the engine rating. The injection system char- acteristics include inadequate or excess penetration, unsuita- ble droplet sizes, and excessive duration of injection, second- ary injection and improper dispersion atomization.
The quality of fuel affects the white smoke produce in an en-
gine. In general, more volatile fuel gives less smoke than heav-
ier fuels of similar cetane number, high cetane number and high volatility both improve the cold smoking performance of an engine. A cetane number of 45 wil gives maximum ac- ceptable white smoke. The cetane number has no effect on production of black smoke.
c) Causes of CO:
it is a product of incomplete combustion due to insufficient amount of air in the air fuel mixture. It is not formed in large quantities due to excess amount of oxygen available during combustion and generally not the concern.
d) Causes of Hydrocarbons:
The pattern of hydrocarbons emission is closely related to many design and operating variables. Two of the important design variables are induction system design and combustion chamber design. The design of the combustion chamber is important in that in the combustion chamber portions of the air fuel mixture which come in direct contact with the cham- ber walls are quenched and do not burned some of this quenched fuel air mixture is forced out of the chamber during exhaust stroke and because of the high local concentration of
IJSER © 2013 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 4, Issue 6, June-2013 3012
ISSN 2229-5518
hydrocarbons in the mixture contributes to the high hydrocar- bon exhaust from the engine.
2.3 Effect of Pollutants
Some of the oxides of nitrogen are very toxic and harmful. The combination of HC and NOx, in the presence of sunlight and certain atmosphere condition produces photochemical smog. NOx, can react with the atmosphere to acidic compounds as well as low level ozone (O3) an irritant. Smoke is a poly ben- zeniod substance, which can cause lung cancer. It has a free valancy available, so it has tremendous agglomeration proper- ties and can be absorbed in metal surfaces, i.e, it is a powerful abruption agent. CO is deadly poisonous gas for both human and environment. Hydrocarbons play an active part in the formation of smog.
2.4 Emission Control Technologys
An emission control program aims at reduce the concentration of CO, HC anf NOx in the exhaust. The main approaches which have been used for this purpose are:
i) Engine Design Modification
The engine modification approach for improving the exhaust emission is aimed all following;
a) Use of leaner air fuel ratios b) Retarding ignition liming
c) Modification of combustion chamber configuration to re- duce quench areas
d) Lower compression ratios
e) Reduce valve overlap
f) Alternation in induction system.
ii) Treatment of Exhaust Gas
Exhaust gas from the engine manifold is treated to reduce HC/CO emission a number of devices have been used. They are given below:
a) Exhaust gas recirculation b) Water injection
c) Catalytic converter
iii) Fuel Modification
a) Compressed natural gas (CNG)
b) liquefied natural gas (LNG)
c) Aqueous salt solution (ASS)
d) Oxygenated fuels such as alcohols, ethers, carbonates, ace-
tates, glycols and esters
3 PROCEDURE AND EXPERIMENTAL SETUP
The experiment is conducted on a 4-stroke diesel engine. The specification of the test engine is given in Appendix-A. The measured parameters through the experiment are rpm, fuel consumption; exhaust temperature etc. Revolution per minute
is reported with the help of tachometer directly indication. The load on engine is determined by pulley belt method in- troducing a pulley to the output shaft and by means of a belt.

The one end of the belt is connected to pulley while another is with spring load where load is found. The rate of fuel con- sumption is determined by measuring time by stopwatch for every 20ML fuel consumption. For this fuel tank is avoided to convey fuel to the plunger, instead of it another pipe is used to the plunger to ensure 20ML for every speed. Exhaust gas temperature is measured by using thermometer.
Fig. 2. Photographic view of Experimental Setup
3.1 Preparation of Salt Diesel Emulsions
The procedure for preparing the salt solution diesel emul- sions is given below.
(i) At first 1% of the emulsifying agent (surfactant TWEEN 80)
is added to the neat diesel fuel and stirred well in a mechani- cal mixer for 15 minutes.
(ii) The salt solution is prepared by mixing
0.2%,0.3%,0.4%,0.5% and 0.6% (sodium carbonate) salt by weight with 10% distilled water
(iii) The salt solution is then added with neat diesel surfactant blend and stirred well for 30 minutes to obtain a macro emul- sion with larger fuel droplets.
In order to reduce the interfacial tension between the two liq- uid phases (salt solution and diesel) and to form a homogene- ous stable solution surfactant is added. Only non-ionic surfac- tants are suggested for preparing the emulsion for engine ap- plications owing to its nonreactive and non-corrosive nature.
3.2 Test Pricedure
At first diesel fuel is used to run the engine and test is con- ducted at load with variable rpm. For each rpm different read- ing is taken such as time taken to the oil consumption, exhaust gas temperature, water inlet and outlet temperature etc. Am- ple time is allowed at each point for steady state condition to be reached before recording any data. Diesel fuel is changed
IJSER © 2013 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 4, Issue 6, June-2013 3013
ISSN 2229-5518
and mixture of salt-solution diesel emulsion is used to run the engine and also test is repeated at same condition. All the data are taken following the same procedure as taken during the
diesel fuel.
4 RESULT AND DISCUSSION
Performance test of the diesel engine with neat diesel and the salt solution was carried out and evaluated. The engine was first run by using the diesel fuel at varying rpm. For each rpm different reading were taken such as time taken for fuel con- sumption, exhaust gas temperature. Then diesel fuel was changed and the engine was run by the salt solution diesel emulsion. All the data were following the same procedure as taken for diesel fuel. All these data are presented in appendix- B and after analysis the data the graph are plotted below
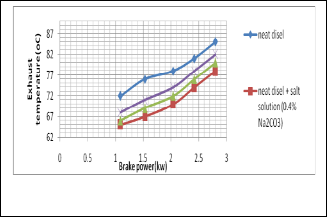
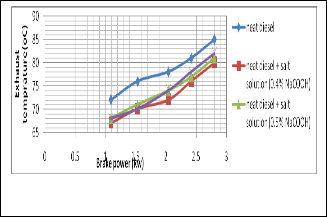
Fig. 5. Shows the change of exhaust temperature with the change of brake power
It is seen that the temperature is gradually increased with brake power. But it is comparatively lower than neat diesel by
using salt diesel emulsion because water diesel emulsion re- duces the temperature of the combustion region.
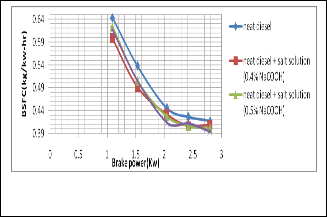
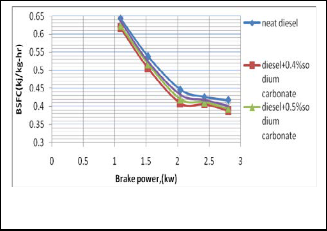
Fig. 3. Shows the change of BSFC with the change of brake power
The figure 3 shows BSFC vs brake power. It is seen that BSFC
is reduced by using salt diesel emulsion. At lower brake pow-
er the BSFC is higher owing to the increasing fuel waste.
Fig. 6. Shows the change of BSFC with the change of brake power
It is seen that BSFC is reduced by using salt diesel emulsion.
At lower brake power the BSFC is higher owing to the increas- ing fuel waste.
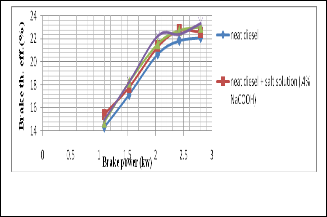
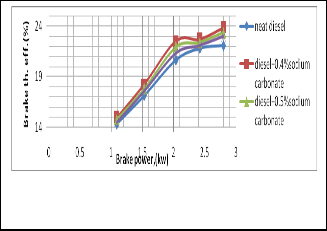
Fig. 4. Shows the change of Brake thermal efficiency with the change of brake power
It is seen that the brake thermal efficiency is increased as the
BSFC is reduced significantly for using salt diesel emulsion.
Fig. 7. Shows the change of Brake thermal efficiency with the change of brake power
IJSER © 2013 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 4, Issue 6, June-2013 3014
ISSN 2229-5518
It is seen that the brake thermal efficiency is increased as the
BSFC is reduced significantly for using salt diesel emulsion
Fig. 8. Shows the change of Exhaust temperature with the change of brake power
It is seen that the temperature is gradually increased with brake power. But it is comparatively lower than neat diesel by using salt diesel emulsion because water diesel emulsion re- duces the temperature of the combustion region.
5 CONCLUSION
The following conclusions are presented from the experi- mental work on salt solution-diesel emulsions with compared to neat diesel:
a) BSFC is decreased maximum up to 8.3% by using salt solu-
tion diesel emulsion compared with neat diesel.
b) Brake thermal efficiency is increased maximum up to 6.08%
by using salt solution diesel emulsion compared with neat
diesel.
c) Exhaust temperature is reduced maximum up to 11.84% by
using salt solution diesel emulsion compared with neat diesel. d) Among all salt solution diesel emulsions, sodium carbonate diesel emulsions is more efficient in improving various per- formance of diesel engine compared to neat diesel.
The most important thing is there is no indication of control-
ling emission in this experiment, because due to unavailability of exhaust gas analyzer. Without this instrument there is no other way to measure NOx. But there is a complete indication of reduction of exhaust gases because there is a significant change in various parameters of diesel engine like reduction in BSFC, increment of break thermal efficiency etc. by using salt solution diesel emulsion. So surely there is some sort of im- provement in exhaust gases. So from above experiment it must be said that reduction of NOx has happened, practically it is found that NOx is reduced up to 66% using salt solution diesel emulsion.
REFERENCES
[1] Mathur, M. L., and Sharma, R.P, “Internal Combustion Engine", PP-
728-731,739-745, 8th edition, 2003
[2] Henry c. perkins, ”Air pollution”, PP-156, 1997
[3] Miwa k., Takeuchi H.,Eguchi T.and Yang C. L.,”Reduction of NOx and particulate of a High speed direct injection with high squish combustion engine symposium”. Japan. 1997
[4] Mathur, M. L., and Sharma, R. P. “ A Course in Internal Combustion
Engine”, Dhanpat Rai Publications, 7th edition pp. 770-774
[5] Henein, N. A, “Analysis of pollutant formation and control and fuel economy in diesel engine”, Progress in energy and combustion science volume 1,1975.
[6] Tadashi Marayaman et.al., “Experimental Reduction of Nox, Smoke and BSFC in a diesel engine Using Uniquely produced water (0-80%) to Fuel emulsion”, SAE: 780224
[7] Nobora Miyamoto and Hideyuki Ogawa “Simultaneous Reduction in Diesel Nox and smoke emissions with aqueous metal-salt solutions directly injected into the combustion chamber”,
[8] Andrews G.E., “The reduction in diesel particulate emission using emulsified fuel”, SAE: 880348
[9] Roy, M M. ,“Exhaust odor reduction in diesel engine fuel-effect of engine pa-
rameters, fuel properly and oxidation catalyst”, Ph. D. thesis, Kitami institute of technology, Japan
[10] Ingram, Colin, “Diesel exhaust odor of small, high speed engine”, SAE paper
780114(1978)
IJSER © 2013 http://www.ijser.org







