International Journal of Scientific & Engineering Research, Volume 2, Issue 12, December-2011 1
ISSN 2229-5518
Comparative analysis of mint gas with R-12 and
R-134 refrigerants
Ratnesh sahu
—————————— ——————————
Human being are looking for ways to keep their food fresh, and found out coldness can satisfy. Therefore the idea of refrigeration was born. For centuries people relay on ice and snow for the purpose of cooling things. Since the Roman Empire, slaves used terracotta pots fanning in water to cool down the food. That is the method of cooling by extracting heat. Until 1844, Jacob Perkins, an American inventor acquired the pattern of the first evaporative cooling refrigerator and a new chapter of refrigeration has begun. After the invention of the first refrigerator, people started to gain more and more interest in using mane-made machines rather than natural ice for cooling food. The early refrigerator models in the nineteenth century made the foundation of the more functional and more stylized refrigerators in the future. Many kinds of refrigerator exist in our society today, each with its own distinct function. But the refrigerator in our home is the most commonly seen and utilized. Many families equip with a refrigerator. No matter of its color size or layout. It serves primary function to keep our food fresh. This paper discuss specifically on home refrigerators
Home refrigerators are a home at low appliance, usually closet shape container with doors used to store food and reduce food bacteria growth by using vapor compression cooling. Vapor compression
A typical home refrigerator consists of these five basic parts- Compressor
Condenser
Expansion valve
Evaporator
The phenomena of green house effect are essential to maintain the temperature of earth. But human activities which are continuously
changing the concentration of atmosphere increasing amount of heat trapping gases and thus earth’s temperature. This effect is known as global warming.
The R-11 is a synthetic chemical product which can be used as a refrigerant. It is stable, non-flammable and non-toxic. It is considered to be a low pressure refrigerant. It has a low side pressure of 0.202 bar at-15C and high pressure of
1.2606 bar at 30C. The latent heat at-15C is 195 KJ/kg. The boiling point at atmospheric pressure is 23.77C. due to its low operating pressures, this refrigerant is exclusive used in large centrifugal compressor of 200TR and above. the leaks may be detected by using a soap solution, a halide torch or by using an electronic detector.
The R-12 is a very popular refrigerant. It is a colorless, almost odorless liquid with boiling point of -29C at atmospheric pressure. It is non-toxic, non-corrosive, non- irritating and non-flammable. It has a relatively low latent
IJSER © 2011
International Journal of Scientific & Engineering Research, Volume 2, Issue 12, December-2011 2
ISSN 2229-5518
heat value which is an advantage in small refrigeration machines. R-12 has a pressure of 0.82 bar at -15C and a pressure of 6.4 bar at 30c. The latent heat of R-12 at -15c is
159KJ/kg.
The preferred replacements of R-12 can be the HFC refrigerants R-134a. This has a boiling point of -26.2C which bears reasonable comparison with the boiling point of R-12 (-29.8C). R-134 is a not a drop in replacement of R-
12 because the refrigerating effect is slightly different. It does not seem to be compatible with conventional lubricants or more winding insulation. It gives higher benefits thenR-12 in using in conventional refrigerators where reasonable condensing temperature is specified. This would appear to be non-flammable and non toxic substitute for R-12 at extreme pressure ratios.
Mint gas is an azeotropic mixture of propane (R290) & iso butane (R600a).it has property very similar to R12 & R 134 which is commonly used refrigerant now a days. This blend of hydrocarbons is used in most of the ac of European cars. It contain 60% propane+40%iso butane.It is named as mint gas because it has cooling property like mint. Moreover it has zero ozone depletion potential and a negliable global warming potential (the two property due to which we need to replace the CFC’s).
This blend is used for domestic refrigerators because of its following reasons-
Operates at similar pressure to R-12 & R-134.
Posses similar volumetric refrigerating effect to R-
12 and R-134a.
Can be used in a R-12 or R-134a compressor.
Can be used with R-12 or R-134a heat exchangers and expansion devices.
Compatible with most common refrigeration
materials and lubricants.
R-290 Propane (C3H8)
Propane is a three-carbon alkane, normally a gas, but compressible to a transportable liquid. A by-product of natural gas processing and petroleum refining, it is commonly used as a fuel for engines, oxy-gas torches, barbecues, portable stoves and residential central heating.
A mixture of propane and butane, used mainly as vehicle fuel, is commonly known as liquefied petroleum gas (LPG or LP gas). It may also contain small amounts of propylene and or butylene. An odorant such as ethanethiol or thiophene is added so that people can easily smell the gas in case of a leak. Boiling point of propane is −187.7 °C, 85.5
K. Propane is generally stored and transported in steel
cylinders as a liquid with a vapor space above the liquid. The vapor pressure in the cylinder is a function of temperature.
Product | Mint gas | R12 | R134a | |||
Chemical type | HC | CFC | HFC | |||
Composition | Azeotropic mixture | Pure | Pure | |||
Ozone depletion potential | 0 | 0.9 | 0 | |||
Global warming potential | 3 | 10600 | 1600 | |||
Normal boiling point | -31c | -30c | -26c | |||
Latent heat | 367 KJ/Kg | 145 KJ/kg | 189 KJ/kg |
IJSER © 2011
International Journal of Scientific & Engineering Research Volume 2, Issue 12, December-2011 4
ISSN 2229-5518
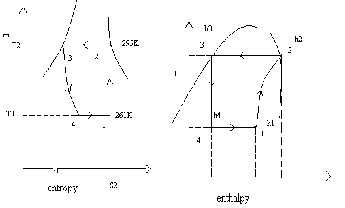
C.O.P from the above-5.15
Taking the case of dry saturated vapors after compression in VCR system

Taking R-134 in above case
Saturation temperature | Pressure Bar | Enthalpy (liquid) KJ/kg | Enthalpy (vapours) KJ/kg | Entropy (liquid) KJ/kg | Entropy (vapours) KJ/kg |
295K | 5.91 | 151.96 | 293.29 | 0.554 | 1.0332 |
261K | 1.39 | 56.32 | 322.58 | 0.226 | 1.2464 |
C.O.P from the above- 4.36
Taking Mint Gas in above case
Saturation temperatu re | Pres sure Bar | Enthalpy (liquid) KJ/kg | Enthalpy (vapours) KJ/kg | Entropy (liquid) KJ/kg | Entropy (vapours ) KJ/kg |
295K | 5.91 | 255.9 | 595.6 | 1.515 | 2.3605 |
261K | 1.39 | 170.45 | 551.8 | 0.8005 | 2.391 |
IJSER © 2011
International Journal of Scientific & Engineering Research Volume 2, Issue 12, December-2011 4
ISSN 2229-5518
In the above study comparison of mint gas is done with r-
12 and r-134 for in domestic refrigerators. From the observation we found that mint gas can be an option which could produce better results. Al though its implementation requires a detail experimental calculations. Mint gas is providing more COP then ordinary refrigerants another advantage of this refrigerant was that it does not react with compressor oil. The only disadvantage associated with this gas is its flammability, which can be an obstacle in its implementation. This problem can be solved by proper design of the refrigerator.
1. United nation environment programme (UNEP) report on refrigeration, air-conditioning and heat pumps.
2. Exergy analysis of R-12 and R-413a. By- Remi
Revellin, Jocelyn Bonjour
3. Exergy analysis and refrigeration effect on the operation and performance limits of a one stage vapour compression refrigeration system. By-camelia STANCIU, Adina GHEORGHIAN,Dorin STANCIU,Alexandru DOBROVICESCU
4. Comparative analysis of performance of three ozone friends HFC refrigerants in a vapour compression refrigerator. By-B.O.BOLAJI,M.A.ATKINTUNDE and T.O.Falade.
5. Performance of a finned tube evaporator optimized
for different refrigerants and its effect on system efficiency. By-Piotr A.Domanski,David Yashar,Minsung kim
6. ASHRE thermo physical properties of refrigerants
chapter 20, ASHRE Fundamental, Inc, Atlanta 20 (2001)1-67.
IJSER © 2011